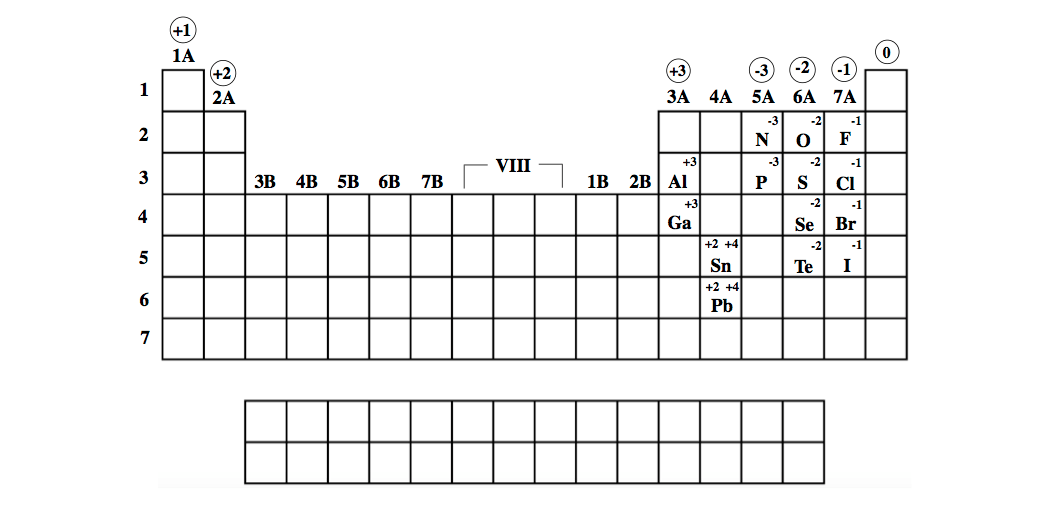 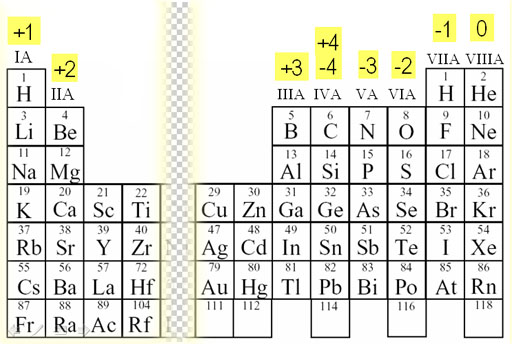
Tin forms stable compounds in both +$ and +2 oxidation states. Germanium forms stable compounds in +4 oxidation states and only few compounds in +2 oxidation states. For example, copper usually has a +1 or +2 valence, while iron typically has a +2 or +3 oxidation state. Metal ions may have other charges or oxidation states. Carbon and silicon mostly show +4 oxidation states. Group I ( alkali metals) carry a +1 charge, Group II (alkaline earths) carry a +2, Group VII (halogens) carry -1, and Group VIII ( noble gases) carry a 0 charge. The valence electronic configuration of these elements is $n$ electrons of valence shell to participate in bonding, heavier elements of the group also have a tendency to show +2 oxidation state. Group IV A of the periodic table includes the elements carbon (C), silicon (Si), germanium (Ge), tin (Sn) and lead (Pb). As you can see from the partial table shown. Carbon of this group shows catenation property and covalency of four. Ions form naturally based on where they are located in the periodic table. They generally have the tendency to form covalent compounds. Transition metals (in the central block between. 
Elements in the same group form ions with the same charge. Recall their valence electronic configuration. The charge of many ions can be worked out using patterns in the periodic table. Hint: The carbon family elements constitute group IV A of the periodic table. The relationship between ionic charge and the number of valence electrons for an element is an essential concept in chemistry. If you look at the periodic table, you might notice that elements on the left side usually become positively charged ions (cations) and elements on the.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
